The role of sexual conflict and sexual selection in trait diversification
We have previously documented the role of sexual conflict in driving the coevolution of sexually antagonistic structures in water striders. We continue to be interested in the pathways connecting microevolutionary processes to macroevolutionary patterns of antagonistic coevolution. We have also begun to study the developmental genes underlying such traits. Through the use of comparative and evo-devo studies, we are exploring the role of sexual conflict and developmental pathways in generating diversity. Water striders exhibit some of the most visible manifestations of sexual conflict, and are an ideal system for answering these questions.

The role of ancient developmental genes in trait diversification. Males of the genus Rheumatobates are known to have highly modified appendages that aid in the grasping of resistant females. The transition from the ancestral to the derived states occurs multiple times and in different ways, leading to a diversity of morphological outcomes (presence and location of antagonistic hooks and spines etc). The remarkable diversity in these antagonistic traits may be driven by two processes – a direct response to differences in female resistance to mating, and/or spatio-temporal variation in the expression of developmental genes, where a variety of outcomes are similarly effective. We have recently uncovered the role of an ancient developmental gene, dll, in controlling the sexually antagonistic modification of male grasping antennae in Rheumatobates rylei. To determine the relative importance of these two alternative processes in generating sexual diversity, we are combining behavioural studies of antennal function with studies examining the role of dll in the diversification of these structures, in Rhematobates spp. that span the range of modified antennae.
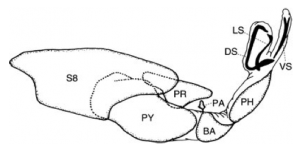
Population level comparative studies of antagonistic coevolution. We have previously quantified the high complexity and rapid shape divergence of genitalia in a comparative study of 15 species of water strider in the genus Gerris. This study also revealed correlative evidence for the roles of both antagonistic selection on the external (grasping) structures of the genitalia, and post-mating sexual selection on the internal structures. Although this work and the work of others suggests sexual selection drives genital diversification, direct evidence is scarce. We are documenting inter-population divergence in genital shape and conducting functional studies of shape variation using 21 populations of G. incognitus from across Canada. We will do so using previously collected behavioural and morphological metrics of antagonism and mating behaviours, population crosses, and the microsatellites developed for Gerris.
The ecological causes and consequences of variation in sexual dimorphism
We are currently working on two projects to explore the ecological forces involved in the evolution of sexual dimorphism.
Ecological factors affecting sexual conflict. We have recently demonstrated an arms race between antagonistic traits at the inter-population level in water striders. We also have an idea of the ecological variables that impact the degree of sexual conflict in water striders (density, sex ratio and food supply), as well as correlative evidence that size dimorphism, which may depend upon food supply, is affecting selection on these antagonistic traits. Employing inter-population surveys, we are now exploring the ecological factors that may affect the position of males and females in this arms race.

The role of ecology and dimorphism in diversification. Sexual dimorphism may be an important component of adaptive radiation if sexually dimorphic species occupy wider niches than monomorphic species, or if sex-specific selection facilitates diversification. To assess the relationship between dimorphism and diversity, we have begun a study of amphibian diversification and its relationship to sexual size dimorphism (SSD). Our initial comparative study of SSD in 940 species has revealed a correlation between SSD in a clade and its species richness. We would now like to understand the ecological factors that mediate sexual dimorphism in this group, and how this dimorphism can in turn facilitate diversification/persistence. We are currently conducting a series of common-garden experiments in replicate, artificial ponds at the Koffler Scientific Reserve, in which we measure sex-specific reaction norms for morphology and life history across ecological gradients that mimic those in the wild, using newts (Notophthalmus viridescens) originating from populations of varying community structure. These experiments will identify the ecological factors that influence the evolution of sexual dimorphism, which in turn is associated with radiation of the lineage.
Selective history and the nature of genetic variance for fitness
We are beginning to investigate how selection history determines the nature of genetic variance in fitness. Our interest arises from two facts: the dependence of key processes such as “good genes” sexual selection on the nature of genetic variance in fitness, and a growing wariness of extrapolating from lab-adapted populations to the wild, given the unique form of selection in the lab.
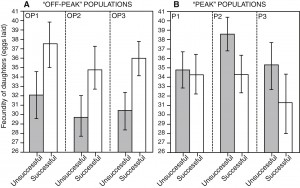
Selective history and the outcome of sexual selection. Good genes models of sexual selection require that natural and sexual selection are concordant and that females can sort among males based on their fitness. If this occurs, favoured males will produce high fitness daughters, and sexual selection will remove maladapted alleles, keeping stable populations on their peak. However, laboratory studies often suggest that fitness variation tends to be sexually antagonistic, with favoured males producing unfit daughters (intralocus sexual conflict). This places natural and sexual selection in opposition. However, the constancy of selection in a homogeneous environment and the absence of any gene flow may exaggerate the importance of sexually antagonistic alleles in laboratory populations. Previous work in our lab suggests that selection history does indeed impact the relative importance of good genes vs intralocus sexual conflict, with good genes effects evident in populations with increased levels of maladaptive variation, and intralocus sexual conflict effects evident in lab-adapted populations. We hope to build on these findings by demonstrating a causal link between adaptation to a constant environment and the loss of sexually concordant fitness variation, by tracking the dynamics of sire-offspring fitness correlations in adapting populations of D. melanogaster through time.
The effect of selective history and environment on the sex-biased transcriptome. Although sex-biased gene expression (SBE) represents a global measure of sexual dimorphism, it is an undeveloped resource for understanding the evolution of sexual dimorphism. Like sexual dimorphism in general, SBE is affected by the environment, but must also reflect a history of sex-biased selection (e.g., sexual selection). However, we do not have good assays of the role of selective history on SBE. To determine the role of sex-biased selection on SBE, we are quantifying the extent of SBE in adapting Drosophila melanogaster populations of differing selection histories. We are also conducting experiments to determine the effect of lab adaptation on SBE, using wild-caught individuals, and assessing the effect of environment and sex-biased behaviours on the extent of SBE.
Evolutionary ecology of life histories
We are interested in the impact of early life conditions on late life outcomes, and the compensatory mechanisms that mediate these outcomes. One of the outcomes we are currently investigating is dispersal. Dispersal plays an important role in community structure and population dynamics. Many early life factors can affect late life dispersal rates, however the effects of multiple factors on dispersal can be complex. Previous work in our lab on backswimmers has shown that predation risk increases dispersal rates among habitats, and that this effect is dose-dependent, with the rate of dispersal increasing with perceived risk. In addition, we have shown that predation risk and backswimmer density interact to affect backswimmer dispersal rates, suggesting that we cannot understand the influence of predation risk without considering other ecological factors. We are also interested in the selective consequence of individual differences in dispersal tendency. If dispersers differ from non-dispersers in genotype or individual condition, then there will be interesting consequences for metacommunity characteristics including persistence and community assembly. We are currently investigating these questions in Notonecta undulata.
